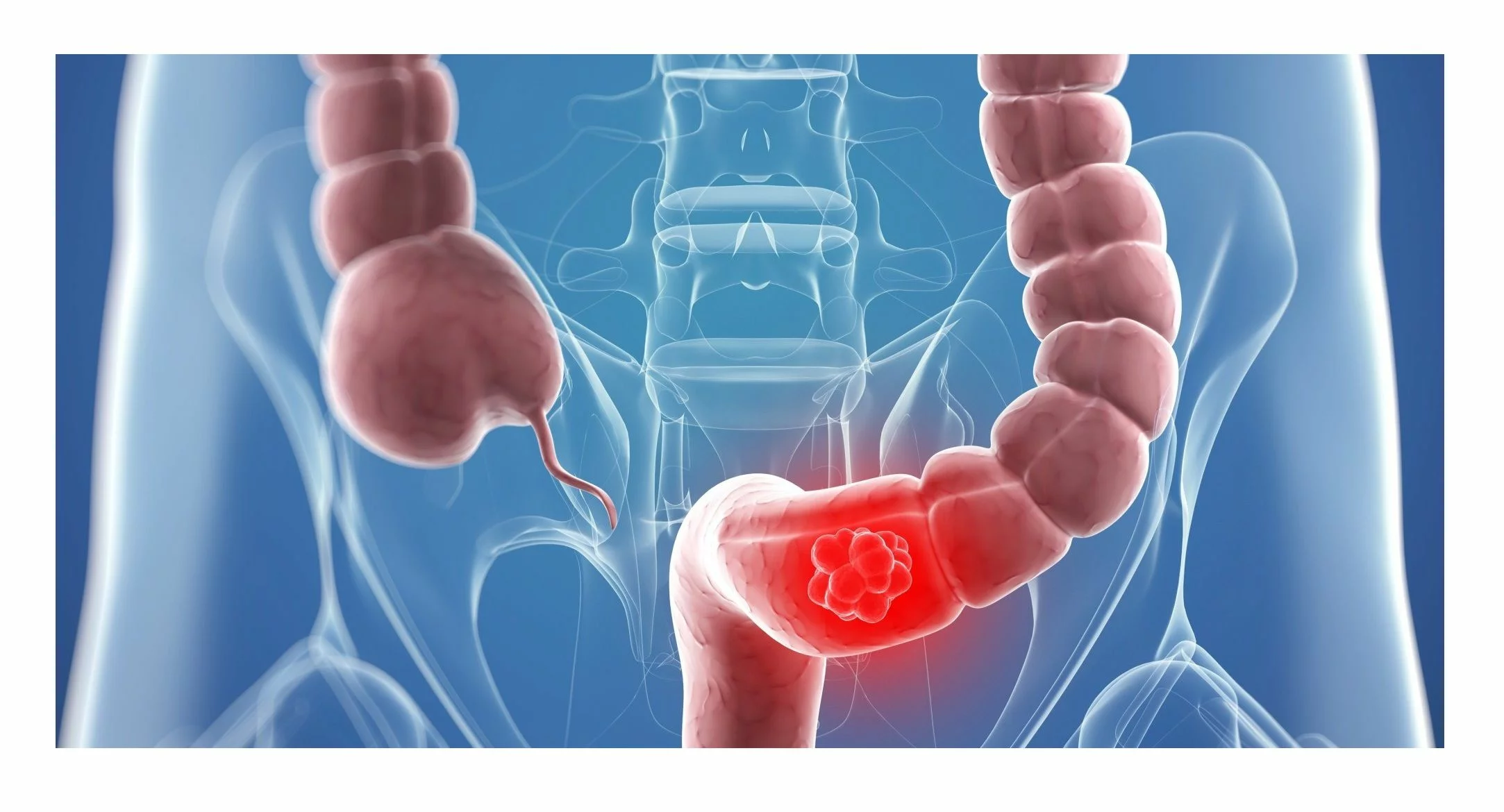
EasyPGX ready FL-DNA (cod. RT029) is the first and only available kit for in vitro diagnostic use which allows the quantitative determination of DNA fragments with dimensions > 200bp present in the faeces*. The quantitative analysis of DNA extracted from the faeces in association with FOBT test, represents a useful tool to improve early diagnosis of CRC allowing to estimate the probability of the CRC presence.
Several studies have shown that the analysis of DNA fragments with dimensions
> 200bp (FL-DNA) allows to identify cancerous lesions. In tumor cells, in which an inhibition of apoptosis occurs, the DNA undergoes a lower degradation and therefore presents fragments > 150-200bp, while in healthy individuals the exfoliated cells undergo apoptosis with consequent DNA degradation in small fragments,.Moreover, in individuals with medium-high hemoglobin levels the evaluation of the amount of FL-DNA allows to identify subgroups with different probabilities of developing a CRC (Colon Rectal Cancer).
Therefore the quantitative analysis of the DNA extracted from the faeces in association with FOBT test, represents a useful tool to improve the early diagnosis of CRC allowing to estimate the probability of presence of CRC.
* The product has been developed under license agreement with Istituto Scientifico Romagnolo per lo Studio e la Cura dei Tumori (IRST) Srl – IRCCS, owner of the following patents:
Molecular early diagnosis of colorectal cancer by quantitative evaluation of DNA extracted from stool: patent number MI2003A 000434. 6988meur.
Method for the identification of colorectal tumors: European patent n. 1601792, 08/12/2010.
Method for the identification of colorectal tumors: United States patent n. 8343722B2, 01/01/2013.
Method for the identification of colorectal tumors: Canada patent n. 59692-NP, 10/01/2013.
